by Jim English
Alpha-lipoic acid (ALA) is a unique, vitamin-like antioxidant that can combat radiation sickness, repair damaged livers, treat diabetes and diabetes-related conditions (polyneuropathy) and protect against oxidative processes that promote premature aging and degenerative diseases. ALA is becoming increasingly recognized as a ‘mitochondrial antioxidant.’ Now a more potent form of ALA – R-lipoic acid – is available to health consumers. Research shows that (R)-lipoic acid is a more biologically active form of ALA that offers greater antioxidant and neuroprotective benefits at substantially lower doses than the synthetic forms of lipoic acid currently available.
Water and Fat Soluble Antioxidant
Although lipoic acid is produced naturally in the body, researchers were virtually unaware of its existence until the 1930s. When pure samples were isolated in the 1950s, ALA was first believed to be a new vitamin. Later, researchers discovered that ALA is, in fact, an essential coenzyme with a vital role in mitochondrial electron transport reactions involved in converting glucose into ATP to produce energy. By 1988 researchers had also learned that ALA is a powerful biological antioxidant, although one with some very unique health properties. What most impressed researchers was the discovery that ALA functions as both a fat and water-soluble antioxidant that can easily cross cell membranes. Thus, ALA can confer free radical protection to both interior and exterior cellular structures.
Regenerating Vitamins E and C
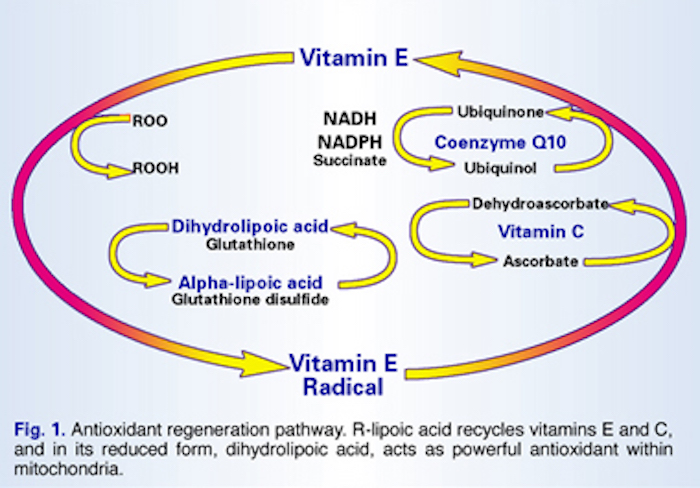
Human aging is marked by a decline in the concentration, synthesis and recycling of central antioxidants, such as vitamin E, vitamin C, co-enzyme Q-10 and glutathione. This loss of antioxidant function seriously impairs the body’s ability to control free radicals. Left unchecked, free radicals – dangerous, unpaired electrons – proliferate throughout the body, damaging cell membranes and organs, impairing immune function, disrupting DNA strands and contributing to the progression of cancer and other degenerative diseases. In recent years researchers have shown how alpha-lipoic acid recycles vitamins E and C to stave off free radical damage.
One of the central components in the antioxidant cycle is vitamin E, a potent biological antioxidant that works to stabilize highly reactive free radicals in lipid (fatty) tissues and membranes (lipoproteins). In the process of quenching fatty free radicals, such as lipid peroxyl and lipid alkoxyl radicals, vitamin E becomes a free radical itself, though one that is far less reactive or damaging than the original radical (Fig. 1).
The vitamin E radical is then regenerated by vitamin C (ascorbic acid). This process recycles vitamin E from a radical back into an antioxidant again, but results in the formation of a new free radical in the form of unstable vitamin C – a semiascorbyl radical. Vitamin C is next recycled by glutathione, a thiol (sulfur-containing compound). Up to this point vitamins E, C and glutathione work in concert to control free radicals and prevent cellular damage. But this is also an important stage where the antioxidant regeneration cycle runs into a limiting factor determined by the availability of glutathione.
Glutathione
Glutathione is the most important member of the thiol group and the body’s primary intracellular antioxidant. In addition to being an important free-radical deactivator, glutathione also plays a vital role in protecting against cataract formation, enhancing immune function, preventing liver damage, slowing the initiation of cancers and eliminating heavy metals. Glutathione is quickly depleted when the body experiences high levels of oxidative stress from causes such as illness, infection, trauma, medication or surgery. Glutathione deficiency is also associated with low protein intake, diabetes, liver disease, cataracts, HIV infection, respiratory distress syndrome, cancer, and idiopathic pulmonary fibrosis.
ALA Boosts Glutathione Levels
Dr. Lester Packer, senior scientist at Lawrence Berkeley Laboratory and head of the Packer Lab at the University of California, has spent over 35 years studying how antioxidants like vitamins C, E and glutathione interact in the body. Dr. Packer first discovered several years ago how vitamin E is ‘recycled’ by vitamin C.
Despite their detailed understanding of the antioxidant regeneration cycle, when Dr. Packer and other researchers tested methods for boosting antioxidant levels they ran into a problem when attempting to increase cellular glutathione levels. Whereas increasing one’s intake of dietary or supplemental sources can readily elevate levels of vitamins E and C, cellular glutathione is only produced in the body.
Glutathione, when taken orally, is broken down in the stomach before reaching the bloodstream. What does end up being absorbed can raise serum levels, but the effect inside of cells is minimal.
This problem was solved when Packer and his team turned to ALA. ‘Alpha-lipoic acid proved to be the missing link,’ Packer said. They discovered that in addition to being a powerful antioxidant in its own right, ALA is able to raise intracellular glutathione levels. ALA is easily absorbed when taken orally, and once inside cells it is quickly converted to its most potent form, dihydrolipoic acid, an even more potent free-radical neutralizer. Because both alpha-lipoic acid and dihydrolipoic acid are antioxidants, their combined actions give them greater antioxidant potency than any other natural antioxidant now known.
The Most Potent Antioxidant
According to Dr. Packer, ALA is more potent than vitamins C and E, and CoQ10, and may, in fact, be the most important antioxidant ever discovered. “The therapeutic potential of alpha-lipoic acid is just beginning to be explored,’ observed Packer, ‘but this compound holds great promise.”
ALA also is important in cell metabolism and is required for production of energy inside cells. Without ALA, cells could not metabolize sugars for energy and would just shut down. This makes alpha-lipoic acid a metabolic antioxidant, able to draw on the cell’s own metabolism to magnify its protective effects and that of other antioxidants.
Experiments carried out by Dr. Packer and colleagues suggest that ALA may also have much broader use in treating other diseases and may have general health benefits when taken as a daily supplement like other antioxidants. ‘Alpha-lipoic acid could have far-reaching consequences in the search for prevention and therapy of chronic degenerative diseases such as diabetes and cardiovascular disease,’ stated Dr. Packer. ‘And because it’s the only antioxidant that can easily get into the brain, it could be useful in preventing damage from a stroke.’
(R)-Lipoic Acid More Effective
Natural ALA, referred to as R-lipoic acid or (R)-ALA, is found in exceedingly miniscule amounts in animal and plant tissues, tightly bound to mitochondrial complexes. Because of the extreme difficulty (and high cost) of isolating natural (R)-ALA, researchers in the US and Europe originally conducted studies with synthetic lipoic acid. Unlike natural (R)-ALA, synthetic lipoic acid contains a 50/50 mixture of two forms (enantiomers) called (R)-ALA and (S)-ALA. Both (R)- and (S)- forms of ALA are isomers – essentially mirror-image molecular formulas, with the atomic arrangements reversed.
Working with synthetic ALA, researchers were able to gain valuable insights into its role as a coenzyme in mitochondrial energy production, and an antioxidant and antioxidant-recycling nutrient. They then began to map out the impressive range of protective health benefits conferred by supplemental ALA. In time researchers had access to pure samples of the natural, biological version, (R)-ALA. They quickly learned that while the body can utilize both forms of ALA, there is a strong preference for the natural, more biologically active (R) form. For example, researchers in Germany reported that, unlike the natural form, (S)-ALA does not improve ATP synthesis in isolated cells. Furthermore, the R-form increased membrane fluidity and transport, while the S-form tended to decrease fluidity.
Conclusion
R-lipoic acid is the biologically active form of the key ‘mitochondrial antioxidant,’ alpha lipoic acid. R-lipoic acid is directly involved in cellular metabolism and is a vital component of the intracellular antioxidant cycle, able to scavenge a variety of free radicals and reactive oxygen species (ROS) while recycling vitamins C, E and glutathione. R-lipoic readily crosses the blood brain barrier and has been shown effective for elevating intracellular glutathione levels. R-LA helps regulate neuronal calcium homeostasis, regulates pro-inflammatory cytokines, and alters the expression of ‘toxic genes.’ R-lipoic acid has been used to treat diabetes and has been recommended as a “neuroprotective agent.” Because R-lipoic is the naturally occurring form found in mitochondrial complexes it offers substantially greater antioxidant and neuroprotective benefits at substantially lower doses than the synthetic forms of lipoic acid currently available.
References
1. Packer L, Tritschler HJ, Wessel K. Neuroprotection by the metabolic antioxidant alpha-lipoic acid. Free Radic Biol Med 1997;22(1-2):359-78.
2. L. Packer, E.H.Witt, H.J. Tritschler, Free Rad Biol and Med 1995; 19: 227-250.
3. Lykkesfeldt J, Hagen TM, Vinarsky V, Ames BN. Age-associated decline in ascorbic acid concentration, recycling, and biosynthesis in rat hepatocytes – reversal with (R)-alpha-lipoic acid supplementation. FASEB J 1998 Sep;12(12):1183-9.
4. Liu J, Atamna H, Kuratsune H, Ames BN. Delaying brain mitochondrial decay and aging with mitochondrial antioxidants and metabolites. Ann N Y Acad Sci 2002 Apr;959:133-66.
5. Liu J, Head E, Gharib AM, Yuan W, Ingersoll RT, Hagen TM, Cotman CW, Ames BN. Memory loss in old rats is associated with brain mitochondrial decay and RNA/DNA oxidation: partial reversal by feeding acetyl-L-carnitine and/or R-alpha -lipoic acid. Proc Natl Acad Sci USA 2002 Feb 19;99(4):2356-61.
6. Hagen TM, Liu J, Lykkesfeldt J, Wehr CM, Ingersoll RT, Vinarsky V, Bartholomew JC, Ames BN. Feeding acetyl-L-carnitine and lipoic acid to old rats significantly improves metabolic function while decreasing oxidative stress. Proc Natl Acad Sci USA 2002 Feb 19;99(4):1870-5.
7. Hofmann M, Mainka P, Tritschler H, Fuchs J, Zimmer G. Decrease of red cell membrane fluidity and -SH groups due to hyperglycemic conditions is counteracted by alpha-lipoic acid. Arch Biochem Biophys 1995 Dec 1;324(1):85-92.
8. Haramaki N, Assadnazari H, Zimmer G, Schepkin V, Packer L. The influence of vitamin E and dihydrolipoic acid on cardiac energy and glutathione status under hypoxia-reoxygenation. Biochem Mol Biol Int 1995 Oct;37(3):591-7. 9. Zimmer G, Beikler TK, Schneider M, Ibel J, Tritschler H, Ulrich H. Dose/response curves of lipoic acid R-and S-forms in the working rat heart during reoxygenation: superiority of the R-enantiomer in enhancement of aortic flow. J Mol Cell Cardiol 1995 Sep;27(9):1895-903.
10. Haramaki N, Packer L, Assadnazari H, Zimmer G. Cardiac recovery during post-ischemic reperfusion is improved by combination of vitamin E with dihydrolipoic acid. Biochem Biophys Res Commun 1993 Nov 15;196(3):1101-7.
11. Moini, H., Tirosh, O., R-Alpha Lipoic Acid Action on Cell Redox Status, the Insulin Receptor, and Glucose Uptake in 3T3-L1 Adipocytes; Archives of Biochem & BioPhys 397, No2 384-391 (2002).
12. Liu, J. Killilea, D.W. et.al., Age-associated mitochondrial oxidative decay: Improvement of carnitine acetyltransferase substrate binding affinity and activity in brain by feeding old rats acetyl-L-carnitine and/or R-alpha-lipoic acid. Proc Nat Acad Sci 99, 1876-1881 (2002).
13. Hager, K., Marahrens, A.,et.al. Alpha lipoic acid as a new treatment option for Alzheimer type dementia, Arch Geron Geriatr 32 (3): 275-282 (2001).
14. Hermann, R.; Niebch, G.; Enantioselective pharmacokinetics and bioavailability of different racemic alpha lipoic acid formulations in healthy volunteers. Eur J Pharm Sci 4: 167-174 (1996).













[…] Your adrenals and immune system will love this product! Refrence: click here […]
[…] It has a half-life of 3-4 hours. The Chelation protocol we follow suggests taking multiple sequential doses every 3 hours for a period of time called a Round. The specific additional supplementation it requires is Vitamin C, E, Magnesium and Zinc . That makes great sense when I ran across this valuable article. Dr. Lester Packer, senior scientist at Lawrence Berkeley Laboratory and head of the Packer Lab at th… -nutritionreview.org […]
[…] (01,02) NutritionReview.com(R)-Lipoic Acid. (source) […]