Maximizing Your Body’s Performance
By Ward Dean, MD and Jim English
Production and management of sustainable biological energy resources is of vital concern for everyone. Disruptions in the normal production of mitochondrial energy can contribute to a wide range of metabolic disturbances and symptoms, including fatigue, immune system dysfunction, dementia, depression, behavioral disturbances, attention deficiency, muscle weakness and pain, angina, heart disease, diabetes, skin rashes, and hair loss. These symptoms of metabolic impairment are also present in persons suffering from acquired diseases, such as Alzheimer’s disease and Chronic Fatigue Syndrome (CFS), and in those with inherited mitochondrial diseases, such as mitochondrial myopathy.
As these conditions share a common link in mechanisms of metabolic energy production, they may also benefit from nutritional strategies that optimize energy production and metabolic pathways.
The Krebs’ Cycle
All cells must produce energy to survive. Hans A. Krebs first elucidated the process of cells converting food into energy, the Citric Acid Cycle, in 1937. Krebs proposed a specific metabolic pathway within the cells to account for the oxidation of the basic components of food – carbohydrates, protein and fats – w for energy. The Krebs’ cycle takes place inside the mitochondria or ‘power plant’ of cells and provides energy required for the organism to function.
Mitochondria are found in all cells in the human body, with the exception of mature red blood cells. The primary function of these tiny organelles (each cell contains between 500 and 2,000 mitochondria) is to convert energy found in nutrient molecules and store it in the form of adenosine triphosphate (ATP). ATP is the universal energy-yielding molecule used by enzymes to perform a wide range of cellular functions. Humans cannot survive, even for a second, without a constant supply of ATP.
In order to carry out energy conversion, mitochondria require oxygen. The purpose of our respiratory and circulatory systems is to deliver oxygen to the tissues for use by mitochondria, and to eliminate carbon dioxide. The consumption of oxygen by mitochondria is called cellular respiration.
In simple terms, the Krebs’ cycle metabolizes acetyl coenzyme A into citric acid and then runs through a complex series of biological oxidations, producing free hydrogen ions. A net of two molecules of ATP is created at this stage in the Krebs’ cycle. The hydrogen ions then enter a biochemical chain, known as oxidative phosphorylation, which is a highly efficient aerobic energy generator. Oxidative phosphorylation generates 36 molecules of ATP during a sequence of steps that combine hydrogen electrons to molecular oxygen to form water. Therefore, each molecule of citric acid that rotates through the Krebs’ cycle, generates 38 molecules of ATP for tissue fuel. (1)
There are different points where metabolites enter the Krebs’ cycle. Most of the products of protein, carbohydrates and fat metabolism are reduced to the molecule acetyl coenzyme A that enters the Krebs’ cycle. Glucose, the primary fuel in the body, is first metabolized into pyruvic acid and then into acetyl coenzyme A. The breakdown of the glucose molecule forms two molecules of ATP for energy in the Embden Meyerhof pathway process of glycolysis. On the other hand, amino acids and some chained fatty acids can be metabolized into Krebs intermediates and enter the cycle at several points.
When oxygen is unavailable or the Krebs’ cycle is inhibited, the body shifts its energy production from the Krebs’ cycle to the Embden Meyerhof pathway of glycolysis, a very inefficient way of making energy.
As well as producing far less energy, glycolysis also produces lactic acid as a byproduct. Increased lactic acid is a common acidotic condition that can be caused by a variety of metabolic problems. Accumulation of lactic acid in muscle tissue produces the pain and inflammation we experience after exercising. While untrained individuals have a low lactate threshold, highly trained, elite athletes are extremely efficient at converting lactate to glucose and therefore have lower lactate levels. (2,3)
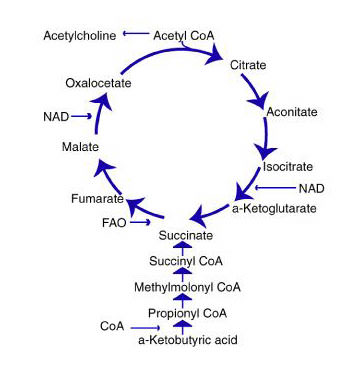
Step 1 The acetic acid subunit of acetyl CoA is combined with oxaloacetate to form a molecule of citrate. Acetyl coenzyme A acts only as a transporter of acetic acid from one enzyme to another. After Step 1, the coenzyme is released by hydrolysis to combine with another acetic acid molecule and begin the Krebs’ Cycle again.
Step 2 The citric acid molecule undergoes an isomerization. A hydroxyl group and a hydrogen molecule are removed from the citrate structure in the form of water. The two carbons form a double bond until the water molecule is added back. Only now, the hydroxyl group and hydrogen molecule are reversed with respect to the original structure of the citrate molecule. Thus, isocitrate is formed.
Step 3 The isocitrate molecule is oxidized by a NAD molecule. The NAD molecule is then reduced by the hydrogen atom and the hydroxyl group. The NAD binds with a hydrogen atom and carries off the other hydrogen atom leaving a carbonyl group. This structure is very unstable, so a molecule of CO2 is released, creating alpha-ketoglutarate.
Step 4 In this step, coenzyme A, returns to oxidize alpha-ketoglutarate. A molecule of NAD is reduced again to form NADH and leaves with another hydrogen. This instability causes a carbonyl group to be released as carbon dioxide and a thioester bond is formed in its place between the former alpha-ketoglutarate and coenzyme A to create a molecule of succinyl-coenzyme A complex.
Step 5 A water molecule sheds its hydrogen atoms to coenzyme A. Then, a free-floating phosphate group displaces coenzyme A and forms a bond with the succinyl complex. The phosphate is then transferred to a molecule of ADP to produce an energy molecule of ATP. It leaves behind a molecule of succinate.
Step 6 In this step, succinate is oxidized by a molecule of FAD (Flavin Adenine Dinucleotide). The FAD removes two hydrogen atoms from the succinate and forms a double bond between the two carbon atoms to create fumarate.
Step 7 An enzyme adds water to the fumarate molecule to form malate. The malate is created by adding one hydrogen atom to a carbon atom and then adding a hydroxyl group to a carbon next to a terminal carbonyl group.
Step 8 In this final step, the malate molecule is oxidized by a NAD molecule. The carbon that carried the hydroxyl group is now converted into a carbonyl group. The end product is oxaloacetate which can then combine with acetyl-coenzyme A and begin the Krebs’ Cycle all over again.
Amino Acid Energy Boosters
Amino acids can be converted via the Krebs’ cycle to glucose for energy or for storage as glycogen and fat. During times of increased stress due to trauma, exercise, starvation and disease states, amino acids can be catabolized into intermediates to produce energy for muscular contraction. This is why taking free form amino acids can boost energy levels and help to prevent hypoglycemia. Additionally, low levels of particular Krebs’ cycle factors may diminish amino acid availability and require supplementation to generate energy and correct metabolic dysfunction. (4-6)
Krebs’ Cycle Shutdown
Under normal conditions all the reactions in the Krebs’ cycle proceed smoothly and ATP is generated without the excessive production of any harmful byproducts. However, different conditions can alter Krebs’ cycle chemistry, causing it to shut down normal energy production. For example, ifcatabolic pathwayssuch as stress, illness, or the synthesis of amino acids consume vital intermediary substances, then the Krebs’ cycle can come to a grinding halt. Interconversions of Krebs’ cycle intermediates are controlled by enzymes that often require vitamin-derived cofactors and minerals to operate. For example, pyruvate is the anaerobic breakdown product of glucose. Its further conversion to acetyl-CoA requires cofactors derived from thiamin, riboflavin, niacin, lipoic acid, and pantothenic acid. When these nutrients are deficient, problems result that disrupt mitochondrial energy production. (7)
In some individuals, a deficiency in the enzyme activity of the Krebs’ cycle may occur when Krebs’ cycle enzymes are bound up by various xenobiotics or endotoxins. The inability of pyruvic acid to enter into the cycle for energy production can shut down the second half of the Krebs’ cycle and create a lactic acid buildup. With increased acidification comes a localized decrease in oxygenation of the tissues, leading to muscle fatigue and other characteristic symptoms.
Researchers have concluded that prolonged exercise to fatigue results in carbohydrate and glycogen depletion that reduces levels of three-carbon compounds from glycolysis. This produces an energy deficiency as the Krebs’ cycle shuts down and lowers aerobic capacity. (8)
Mitochondrial Insufficiency, Alzheimer’s and CFS
A deficiency in one or more Krebs’ cycle intermediates and an inhibition of normal energy production may cause a wide range of metabolic disturbances and symptoms. A deficiency of malic acid and fumaric acid is linked to chronic fatigue and psoriasis. Disturbances in mitochondrial energy production contribute to a variety of neurological and physical problems. Impaired oxidative and energy metabolism are indicators of Alzheimer’s disease. These disturbances of energy production can create abnormal spilling of Krebs’ cycle byproducts into the urine. (9-13)
Chronic Fatigue Syndrome (CFS) represents a condition of debilitating fatigue. Some neurological symptoms of CFS are poor attention, memory loss, lack of concentration and depression. An underlying cause of CFS may be an impairment in the production of mitochondrial adenosine triphosphate (ATP), the fundamental cellular energy source. Studies have found that CFS patients have elevated blood levels of lactate, indicating suboptimal aerobic ATP production that can lead to fatigue and muscle aches.
Studies have shown administering specific Krebs’ cycle amino acid precursors and intermediates to stimulate energy production significantly reduce symptoms of CFS. Therefore supplying a complete range of Krebs’ cycle factors and lactic acid-buffering agents may be of great benefit to the sufferers of CFS. (14,15)
Fatigue, Acidity and Exercise
Intense exercise involves an anaerobic component that can lead to a significant reduction of ATP, a buildup of lactic acid, and an increase in tissue acidity. Acidity can normally be buffered by the body, but high levels of physical stress can rapidly produce large quantities of lactic acid and overwhelm the body. The resulting excess acidity may lead to premature muscle fatigue.47 A buildup of lactic acid occurs whenever inadequate supplies of oxygen create a hypoxic condition, preventing the complete aerobic metabolism of glucose. As a result, ATP cannot be generated in adequate amounts, causing skeletal muscles to weaken, become fatigued and less efficient at contracting.
In addition, the accumulation of large amounts of metabolic anaerobic byproducts can lower intracellular pH, inhibit muscle contraction, and may cause acidosis. The accumulation of metabolic byproducts, like hydrogen ions, interferes with muscle contractions and ATP energy release. Metabolic fatigue from exercise occurs when the muscle’s need for ATP has outstripped production capacity. Chronic acidosis in muscle tissue causes a negative nitrogen balance and loss of muscle protein. (16-23), (48-54)
Counteracting Fatigue
There are several ways to counteract metabolic fatigue. One approach is to increase the production of ATP by eating correctly and taking supplements that stimulate the pathways that make ATP. There are three pathways used by the body to produce energy: (1) the immediate energy-producing ATP-PC system; (2) the short-term anaerobic energy system of glycolysis; and (3) the long-term aerobic energy system of oxygen. Creatine supplements stimulate the ATP-PC system. The Krebs’ cycle factors, their precursor amino acids and food metabolites enhance glycolysis and aerobic energy production.
Anti-Fatigue Buffers
An entirely different way to relieve fatigue is by buffering hydrogen ions and lactic acid. Buffers can prevent intracellular acidosis from producing fatigue and muscle breakdown. The intracellular buffers that have been shown to improve buffering capacity and enhance sports performance are phosphates, carnosine and bicarbonates. While bicarbonate is the main blood buffer, it plays a minor role in muscle tissue buffering. Phosphates and the amino acid carnosine account for 90% of muscle buffering. (24,55)
Potassium and Sodium Phosphate
The alkaline salts of phosphor act as buffers to improve athletic performance in several ways. First, they reduce lactic acid buildup and intracellular acidosis to delay muscle fatigue. Secondly, phosphate supplements increase the concentration of 2,3-DPG (diphosphoglycerate) in red blood cells which speeds the release of oxygen from hemoglobin to muscles. Lastly, phosphate supplements help in the phosphorylation of creatine to creatine phosphate to reform ATP and increase energy.
Numerous studies spanning decades have demonstrated the benefits of phosphate blood buffer supplements on improving athletic performance. The early use of phosphates to improve physical performance began with Embden (of the Embden Meyerhof pathway) during World War I. German soldiers were given sodium acid phosphate to reduce battle fatigue. East German researchers found that taking 1 to 3 grams of phosphates one hour prior to workouts improved psychomotor performance and prevented muscle fatigue. More recently researchers at the University of Memphis found that phosphates lead to a 10% increase in VO2max, a 10% increase in maximal oxygen uptake and a 9% increase in power output at anaerobic threshold. (25-30), (56-61)
Krebs’ Cycle Acids
Alpha-ketoglutaric Acid, Malic Acid, Fumaric Acid, Succinic Acid, Citric Acid, Pyruvic Acid, Pantothenic Acid
These acids are intermediate compounds that are found in the Krebs’ cycle and are necessary to generate cellular energy for tissue fuel. Supplementing these essential Krebs’ cycle acids in the presence of nutrient cofactors can enable a partially completed Krebs’ cycle to go to completion. They can prevent and remove the harmful byproducts that are generated from abnormal energy production in the mitochondria. And they can stimulate a high yield of ATP from the mitochondria for tissue energy.
Supplementing these Krebs’ cycle fuel sources may be advisable for different purposes. They can help correct certain metabolic disorders that result from abnormal mitochondria energy production. They can provide an ergogenic edge in athletic performance by generating muscle energy, increasing aerobic capacity and preventing fatigue. They may be even more helpful for improving athletic performance when used in conjunction with alkalizers that buffer lactic acid build-up in muscle tissue and improve tissue oxygenation.
Alpha-ketoglutaric Acid (AKG)
Alpha-ketoglutaric acid plays a vital role in the Krebs’ cycle production of energy. As a precursor of the amino acid, glutamic acid, AKG stabilizes blood glucose levels during exercise. Alpha-ketoglutaric acid benefits the athlete by supporting protein synthesis, allowing for longer, more intense workouts, and by promoting healthy nitrogen balance.
Studies of patients given supplemental alpha-keto-glutarate following surgery found a nitrogen-sparing effect and a reduction in loss of lean body mass. Alpha-ketoglutaric acid helps reduce ammonium levels that may interfere with exercise performance. Studies have demonstrated that ammonia formed in the muscle, kidney and brain combines with alpha-ketoglutarate and L-glutamate to reduce ammonia toxicity. (31-33), (16-18)
Malic Acid
Malic acid acts as a catalyst in the Krebs’ cycle to increase energy production from the burning of pyruvic acid. Malic acid also aids in exercise recovery by counteracting the buildup of lactic acid. Supplementation of malic acid has been reported to be beneficial in Chronic Fatigue Syndrome by reducing symptoms of persistent fatigue, muscular myalgia and arthritic-like pains.
Fumaric Acid
Fumaric acid is the trans-isomer of malic acid that enters the citric acid cycle. It’s a byproduct at certain stages in the arginine-urea cycle and purine biosynthesis. In healthy individuals, fumaric acid is formed in the skin from exposure to sunlight. A deficiency of fumaric acid leads to the accumulation of metabolic half-products that may be responsible for causing the skin lesions of psoriasis. Sufferers of psoriasis have a biochemical defect in which they do not produce enough fumaric acid, requiring prolonged exposure to the sun. Administration of fumaric acid to individuals suffering from psoriasis has caused a gradual elimination of the symptoms. (40-47), (25-32)
Succinic Acid
Succinic acid, like other Krebs’ cycle intermediates, is an entry pathway for other metabolites into the cycle and is involved in a variety of important biological actions. In addition to its enzyme activity, it combines with protein to rebuild muscle fiber and nerve endings, and helps fight infection. Individuals with Chronic Fatigue Syndrome have shown low levels of succinic acid in their urine.
Several amino acids are metabolized into succinic acid, providing a source of anaerobic and aerobic energy. Amino acids that are metabolized into succinic acid have been shown to be important in supplying the heart with fuel for myocardium contractions under low oxygen conditions. The amino acid GABA can either be oxidized to succinic acid for cellular energy production, or reduced to GHB, depending on the metabolic needs of the body. (48-50), (33-35)
Citric Acid
Citric acid, a natural organic acid present to some extent in all plant and animal tissues, occupies a pivotal location in the Krebs’ cycle. After proteins, fats, carbohydrates and amino acids have been oxidized into acetyl coenzyme A, the acetic acid subunit of acetyl CoA is combined with oxaloacetate to form a molecule of citrate. The acetyl coenzyme A acts as a transporter of acetic acid from one enzyme to another.
First isolated by the German biochemist, Karl Wilhelm Steele in 1784, today citric acid is widely respected for relieving conditions of fatigue, poor digestion, cold and flu infections, asthma, hypertension and cholesterol deposits in blood vessels.
Pyruvic Acid
Pyruvic acid is a three-carbon ketoacid produced in the end stages of glycolysis. In the mitochondria, pyruvic acid is either reduced to lactate in the cytoplasm, or oxidized to acetyl CoA.
Research has shown that taking pyruvate (the salt of pyruvic acid) can increase muscle endurance and promote fat loss. Pyruvic acid also appears to increase the amount of glucose that enters muscle cells from the circulating blood. This ability of pyruvic acid leads to increases in immediate available energy, as well as increasing stored muscle glycogen levels for future energy. Research has shown that pyruvic acid increases muscle endurance and improves cardiac efficiency.
In one study pyruvic acid was found to increase glucose extraction by almost 300% and muscle glycogen by 50% after one hour of exercise. The researchers found that arm endurance increased by 150% and leg endurance by 60%. Another study conducted at the University of Pittsburgh School of Medicine found that pyruvic acid produced a significant amount of weight loss and fat loss in obese women on a low calorie liquid diet. Two potential mechanisms by which pyruvic acid enhances both fat and weight loss are through increasing both resting metabolic rate and fat utilization. (51-56), (36-41)
Pantothenic Acid
Vitamin B5 is required for the synthesis of coenzyme A. Supplementation of panthenine (pantothenate bound to cysteamine) has been shown to reduce elevated blood lipids in humans. It is postulated that this action is due to the accelerated synthesis of coenzyme A. It has also produced an anti-arrhythmic effect in animal hearts by increasing ATP synthesis. A study of elite distance runners who were given two grams of pantothenic acid daily for two weeks found a 17% reduction in lactic acid buildup and a seven percent reduction in oxygen consumption during prolonged, strenuous exercise. (57-61), (42-46)
Summary
The Krebs’ cycle is an eloquent and essential system designed to generate large amounts of cellular energy required for life. Disruption of the Krebs’ cycle, whether caused by deficiencies in energy substrates, acquired or inherited disease states, or physical stress, leads to an inhibition of normal energy production and contributes to a wide range of metabolic disturbances and symptoms.
The use of supplemental Krebs’ cycle acids and anti-fatigue buffers can assist in the management of mitochondrial energy substrates and increase cellular energy production. Such a nutritional approach can be of benefit to athletes, anyone who is aging, as well as those suffering from metabolic disturbances caused by inherited mitochondrial diseases or acquired diseases, such as Alzheimer’s disease and Chronic Fatigue Syndrome (CFS).
References
1. Lehninger AL et al Cox, Principles of Biochemistry W H Freeman & Co. New York, NY 1999
2. McArdle WD et al Exercise Physiology : Energy, Nutrition, and Human Performance 4th edition Lea & Febiger; Philadelphia PA 1996
3. Wilmore JH, David Costill DL, Physiology of Sport and Exercise 2nd edition, Human Kinetics Publishing Champaign, IL 1999
4. Dohm GL; Kasperek GJ et al Protein metabolism during endurance exercise. Fed Proc, 1985 Feb, 44:2, 348-52
5. Shaw D, et al., Management of fatigue: A physiological approach. Am J Med Sci 1962; 243:758.
6. Hicks, JT, Treatment of fatigue in general practice: A double blind study. Clin Med 1964 (Jan.):85.
7. DiPasquale M, Amino Acids and Proteins for the Athlete: The Anabolic Edge. CRC Press Boca Raton Florida 1997
8. Sahlin K; Katz A; Broberg S Tricarboxylic acid cycle intermediates in human muscle during prolonged exercise, Am J Physiol 1995 Feb;268
9. Bralley A and RS. Lord, Organic Compounds in Urine: Metabolic Profiling to Assess Functional Nutrient Deficiencies Gut Dysbiosis, and Toxicity Natural Medicine Pizzorno and Murray ed, Churchill Livingstone, London., 1998
10. Redjems Bennani N et al Abnormal substrate levels that depend upon mitochondrial function in cerebrospinal fluid from Alzheimer patients. Gerontology, 1998, 44:5, 300-4
11. Pisarenko OI; Studneva IM; et al Formation of products of anaerobic metabolism in the ischemic myocardium. Biokhimiia, 1988 Mar, 53:3, 491-7
12. Gabreëls FJ; et al Defects in citric acid cycle and the electron transport chain in progressive poliodystrophy. Acta Neurol Scand, 1984 Sep, 70:3, 145-54
13. Efimov AS; Gulyi MF; Shcherbak AV; Dzvonkevich ND Levels of Krebs cycle metabolites in the blood and urine of patients with diabetes mellitus Probl Endokrinol (Mosk), 1983 Mar, 29:2, 10-4
14. Bralley A Lord RS.Chronic Fatigue and Amino Acids. J. of Applied Nutr., 46(3):74-78(1994)
15. Riley MS, O’Brien CJ, McCluskey DR, Bell NP, Nicholls DP. Aerobic work capacity in patients with chronic fatigue syndrome. Br Med J 1990; 301:953-956.
16. Norman B. Sollevi A, Kaijser L, Jansson E. ATP breakdown products in human skeletal muscle during prolonged exercise to exhaustion. Clin Physiol 1987; 7(6):503-10.
17. Layzer RB. Muscle metabolism during fatigue and work. Baillieres Clin Endocrinol Metab 1990; 4(3):441 – 976.
18. Degroot M, Massie BM, Boska M, Gober J. Miller RG, Weiner MW Dissociation of [H’] from fatigue–human muscle detected by high time resolution 31P-NMR. Muscle Nerve 1993; 16(1):91-8.
19. Mainwood GW, Renaud JM. The effect of acid-base balance on fatigue of skeletal muscle. Can J Phy Pharmacol 1985; 63(5):403-16.
20. Sjogaard G. Role of exercise-induced potassium fluxes underlying muscle fatigue: a brief review. Ca Physiol Pharmacol 1991, 69(2):238-45.
21. Juel C, Bangsbo J. Graham T. Saltin B. Lactate and potassium fluxes from human skeletal muscle during and after intense, dynamic, knee extensor exercise. Acta Physiol Scand 1990; 140(2): 147-59.
22. Reaich D, et al TH. Ammonium chloride-induced acidosis increse protein breakdown and amino acid oxidation in humans. Am J Physiol 1992; 263(4 Pt l):E735-9.
23. Parkhouse WS, McKenzie DC. Possible contribution of skeletal muscle buffers to enhanced anaerobic performance: a brief review. Med Sci Sports Exerc 1984; 16(4):328-338.
24. Cade R. Conte M, Zauner C, et al. Effects of phosphate loading on 2,3-diphosphoglycerate and maxi oxygen uptake. Med Sci Sports Exerc 1984; 16:263.
25. Kraemer WJ, Gordon SE, Lynch JM, Pop ME, Clark KL. Effects of multibuffer supplementation on acid-base balance and 2,3-diphosphoglycerate following repetitive anaerobic exercise. Int J Sport Nutr.
26. Embden, G., Grafe, E., and Schmitz, E., Uber Steigerung der Leist-ungsfahigkeit durch Phosphatzufahr, Z. Physiol. Chem., 113, 67, 1921.
27. Herxheimer, H., Zur Physiologie der maximalen Muskelarbeit in Sport, Z. Physik. Therap., 44, 55, 1933.
28. Keller, W. D. and Kraut, H. A., Work and nutrition, World Rev. Nutr. Diet., 3, 65, 1959.
29. Cade, R., Conte, M., Zauner, C., Mars, D., Peterson, J., Lunne, D., Hommen, N., and Packer D., Effects of phosphate loading on 2,3-diphosphoglycerate and maximal oxygen uptake, Med. Sci. Sports Exer., 16, 263, 1984.
30. Hammarkvist F et al Alpha-ketoglutarate preserves protein synthesis and free glutamine in skeletal muscles after surgery. Surgery 101(1): 28-31,1991
31. Wernerman J Hammarkvist F. Alpha-ketoglutarate and postoperative catabolism. Lancet 335(8691)701-703,1990
32. Marconi C Sassi G The effect of an alpha-ketoglutarate-pyridoxine complex on human maximal aerobic and anaerobic performance. Eur J Appli Phys 49(3) 307-17, 1982
33. St. Claire SM, et al., ‘Diagnosis and Treatment of Fibromyalgia Syndrome,’ J Neuromusc Sys 2(3) (1994): 101-11.
34. Dunaev VV, et al., ‘Effect of Malic Acid Salts on Physical Work Capacity and its Recovery after Exhausting Muscular Activity,’ Famakol Toksikol 51(3) (1988): 1-25.
35. Bobyleva-Guarriero, et al., ‘The Role of Malate in Exercise-induced Enhancement of Mitochondrial Espiration,’ Arch Biochem Biophys 245(2) (19086): 470-76.
36. Russell IJ, et al., ‘Treatment of Fibromyalgia Syndrome with Super Malic: A Randomized, Double Blind, Placebo Controller, Crossover Pilot Study,’ J Rheu.
37. Abraham, G A Management of fibromyalgia: Rationale for the use of magnesium and malic acid. J. of Nutr. Med., 92 (3): 49, 1992
38. Kostin VI A preclinical evaluation of the antianginal activity of energy metabolism intermediates. Farmakol Toksikol, 1989 Nov, 52:6, 49-52
39. Weinstein, G.D. & Frost, P. Abnormal cell profliferation in psoriasis. J. Invest. Derm. 50:254, 1968.
40. Kolbach DN, Nieboer C. Fumaric acid therapy in psoriasis: results and side effects of 2 years of treatment. J Am Acad Dermatol 1992;27:769–71.
41. Cathcart R Perry A et al, The Surprising Psoriasis Treatment. The Arthritis Trust, Franklin, TN. 1990
42. Altmeyer PJ, Matthes U, Pawlak F, et al. Antipsoriatic effect of fumaric acid derivatives. J Am Acad Dermatol 1994;30:977–81.
43. Hagedorn, K. et al. Fumarsauremonoathylester: Wirkung auf DNA-Synthese und erste tierexperimentelle Befunde. Arch. Derm. Res. 254:67-73, 1975.
44. Bruynzeel, D. Kan fumaarzuur worden toegepast bij psoriasis? Ned. Tijdschr. Geneeskd. 128(35):1677, 1984.
45. Baron, D. fumaarzuur worden toegpast bij psoriasis (Vraag en Antwoord). Ned. tijdschr. Geneeskd. 128(24):1152-1153, 1984.
46. Elpeleg, O. N, Amir, N, Christensen, E, Variability of clinical presentation in fumarate hydratase deficiency. J Pediatr 1992: 121: 752-4.
47. Hochachka PW Dressendorfer RH Succinate accumulation in man during exercise. Europ J Appl Physiol 35(4): 235-242, 1976
48. Pisarenko OL et al Formation of intermediate products of the tricarboxylic acid cycle and ammonia from free amino acids in anoxic heart muscles. Biokhimiia 51(8):1276-85.
49. Wiesner RJ et al The anaerobic heart: succinate formation and mechanical performance. Exp Biol 45(1) :55-64, 1986
50. Greenawalt, K, et al. ‘The effect of feeding dihydroxyacetone and pyruvate on resting energy expenditure and postprandial thermogenesis in obese women.’ FASEB (Federation of American Societies for Experimental Biology) JOURNAL. 1988 2(5):ABSTRACT 5371.
51. Robertson, R.J., et al, Greenawalt, K.D. ‘Blood glucose extraction as a mediator of perceived exertion during prolonged exercise.’ European Journal of Applied Physiology 61:100-105.
52. Stanko, R T, et al. ‘Inhibition of regain in body weight and fat with addition of 3-carbon compounds to the diet with hyperenergetic refeeding after weight reduction.’ Int J Obes 1996 20(10):925-930.
53. Stanko, R.T., Tietze, D.L., Arch, J.E. ‘Body composition, energy utilization, and nitrogen metabolism with a severely restricted diet supplemented with dihydroxyacetone and pyruvate.’ Am. J. of Clin. Nutr. 55:771-775, 1992.
54. Stanko, R.T., et al ‘Enhancement of arm-exercise endurance capacity with dihydroxyacetone and pyruvate.’ J. of Applied Phys. 68:119-124, 1990.
55. Stanko, R.T., Teitze, D.L., Arch, J.E. ‘Body composition, energy utilization, and nitrogen metabolism with a 4.25-MJ/d low-energy diet supplemented with pyruvate.’ Am. J. of Clin. Nutr. 56:630-635.
56. Litoff,D., Scherzer, H., and Harrison, J., Effects of pantothenic acid supplementation on human exercise, Med. Sci. Sports Exer., 17, 287, 1985.
57. Branca, D., Scutari, G., and Siliprandi, N., Pantethine and pantothenate effect on the CoA content of rat liver, Int. J. Vit. Nutr. Res., 54, 211, 1984.
58. Hyashi, H., et al Effects of pantethine on action potential of canine papillary muscle during hypoxic perfusion, Jpn Heart J., 26, 289, 1985.
59. Shibano, T. and Abiko, Y., Effect of pantethine on myocardial pH reduced by partial occlusion of the coronary artery in dogs with special reference to the myocardial levels of adenosine triphosphate, creatine phosphate, and lactate, Arch. Int. Pharmacodyn., 255, 281, 1982.
60. Angelico, M., Pinto, G., et al Improvement in serum lipid profile in hyperlipoproteinaemic patients after treatment with pantethine: a ross-over, double-blind trial versus placebo, Curr. Ther. Res., 33, 1091, 1983.
61. Quesch PA, Colliander EB, Kaiser R Muscle metabolism during intense, heavy-resistance exercise. Eur JA, Physiol 1986; 55(4):362-366.












[…] Maximising your body’s performance (the Krebs cycle in relation to fibromyalgia etc.) […]
[…] https://nutritionreview.org/2013/04/krebs-cycle-intermediates/ […]
[…] reduced pain and tenderness in those with fibromyalgia at the right dosage and duration. A deficiency of malic acid and fumaric acid is linked to chronic fatigue and psoriasis, while […]
[…] reduced pain and tenderness in those with fibromyalgia at the right dosage and duration. A deficiency of malic acid and fumaric acid is linked to chronic fatigue and psoriasis, while […]
[…] reduced pain and tenderness in those with fibromyalgia at the right dosage and duration. A deficiency of malic acid and fumaric acid is linked to chronic fatigue and psoriasis, while […]
[…] Krebs’ Cycle Intermediates | Nutrition Review – In order to carry out energy conversion, mitochondria require oxygen. The purpose of our respiratory and circulatory systems is to deliver oxygen to the tissues for … […]
[…] https://nutritionreview.org/2013/04/krebs-cycle-intermediates/ […]
[…] we are concerned about malic acid, our focus is on the Kreb’s cycle. Mitochondria are these tiny units within each cell of the body (except […]
[…] Download Image More @ nutritionreview.org […]
[…] do l-cytruliny, kwas jabłkowy w jabłczanie l-cytruliny bierze udział w cyklu Krebsa ( 12 […]
[…] Sin embargo, a diferencia de la l-citrulina, el ácido málico del malato de la l-citrulina participa en el ciclo de Krebs (12). […]
[…] malic acids come from apples, but we also have them in our bodies as a vital catalyst in the Krebs energy cycle. Ascorbic acid is more commonly known as vitamin C that strengthens the immune system in fighting […]
[…] W Dean, MD, J English, 2013.Krebs Cycle Intermediaries.www.nutritionreview.org […]
[…] body uses to create ATP. Citric acid activates and maintains the cycle, and malic acid acts as a catalyst to maximise ATP […]
[…] However not like l-citrulline, the malic acid in l-citrulline malate participates within the Krebs cycle (12). […]
[…] is actually a key ingredient in that energy creation process we spoke about earlier. It acts as a catalyst to increase the efficiency of the energy production cycle. It’s also one of the better-studied […]
[…] acid acts as a catalyst for the production of ATP, a molecule that carries energy to your cells, and magnesium helps to […]