The mysterious inner workings of Chang Shan – a Chinese herbal medicine used for thousands of years to treat fevers associated with malaria – have been uncovered thanks to a high-resolution structure solved at The Scripps Research Institute (TSRI).
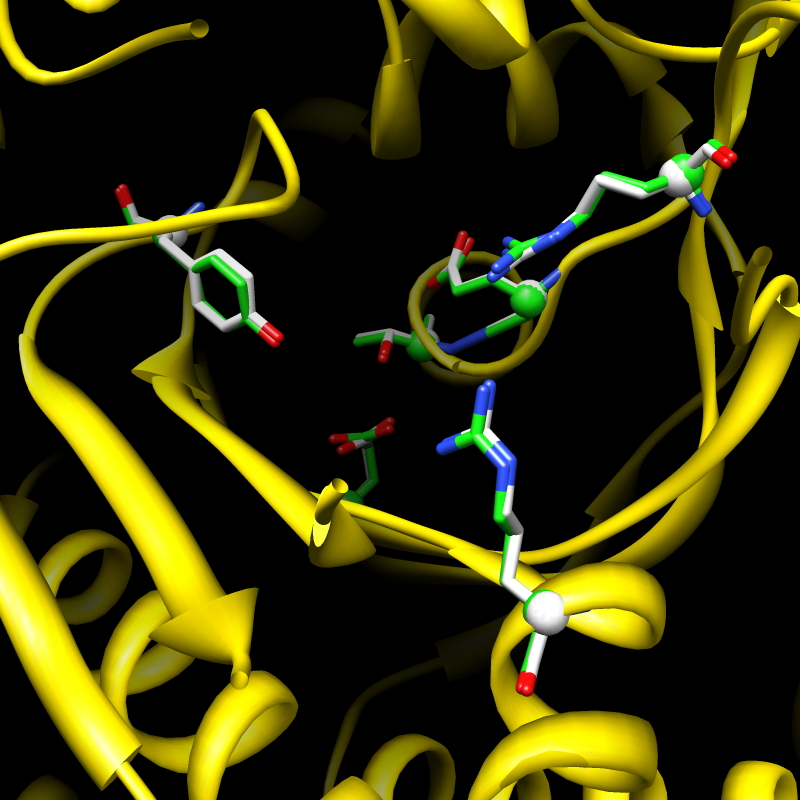 Described in the journal Nature, the structure shows in atomic detail how a two-headed compound derived from the active ingredient in Chang Shan works. Scientists have known that this compound, called halofuginone, can suppress parts of the immune system, but nobody understood exactly how it worked.
Described in the journal Nature, the structure shows in atomic detail how a two-headed compound derived from the active ingredient in Chang Shan works. Scientists have known that this compound, called halofuginone, can suppress parts of the immune system, but nobody understood exactly how it worked.
The new structure shows that halofuginone jams the gears of a molecular machine that carries out “aminoacylation,” a crucial biological process that allows organisms to synthesize the proteins they need to live. Chang Shan, also known as Dichroa febrifuga Lour, probably helps with malarial fevers because traces of a halofuginone-like chemical in the herb interfere with this same process in malaria parasites, killing them in an infected person’s bloodstream.
“Our new results solved a mystery that has puzzled people about the mechanism of action of a medicine that has been used to treat fever from a malaria infection going back probably 2,000 years or more,” said Paul Schimmel, PhD, the Ernest and Jean Hahn Professor and Chair of Molecular Biology and Chemistry and member of The Skaggs Institute for Chemical Biology at TSRI. Schimmel led the research with TSRI postdoctoral fellow Huihao Zhou, PhD.
Halofuginone has been in clinical trials for cancer, but the high-resolution picture of the molecule suggests it has a modularity that would make it useful as a template to create new drugs for numerous other diseases.
Source: “ATP-Directed Capture of Bioactive Herbal-Based Medicine on Human tRNA Synthetase,” by Huihao Zhou, Litao Sun, Xiang-Lei Yang and Paul Schimmel, published in the journal Nature. http://bit.ly/1rQOvMa
Image: Aminoacylation protein translation leading to synthesis of aminoacyl tRNA.