By Jim English
Our fast-paced, interconnected world is seemingly fueled by non-stop, 24-hour news cycles that are a significant source of stress and anxiety. And while a little stress is a good thing (without it life would be uneventful and dull), chronic stress can build up to leave us feeling anxious, worn-out, short-tempered and depressed. These symptoms are manifestations of what is happening inside the body — and in particular, the brain — when we’re stressed. Now scientists are revealing how living with constant fear and anxiety is actively disrupting brain chemistry and damaging brain cells, resulting in physically altered brain structures and accelerated brain aging.
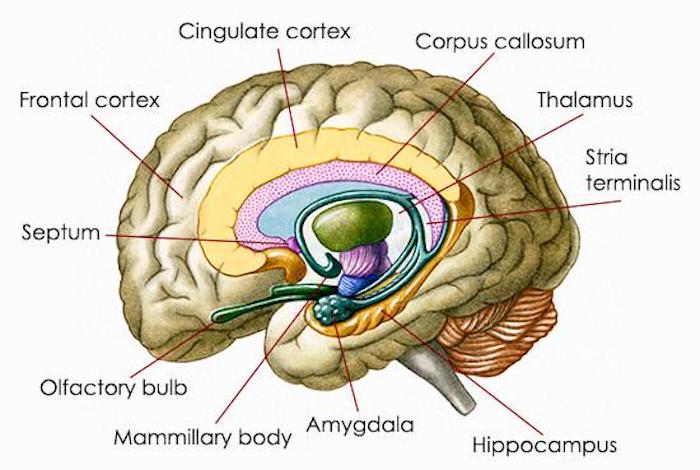
New research has revealed that the outward manifestations of stress are just the tip of the iceberg, with much more going on beneath the surface. In fact, stress has been shown to actually change the brain’s structure. Stress is especially damaging to the amygdala, an almond-shaped region of the brain that regulates basic emotions, such as fear and anxiety. Researchers have known that exposure to prolonged stress can cause structural changes in the amygdala. These changes have been linked to behaviors associated with anxiety and depressive disorders.
But new studies in rats are showing that all it takes is one stressful event to cause delayed and long-term psychological trauma. Many studies investigating the effects of stress on rodents expose the animals to chronic—in other words long-term—stress. In studies where researchers did assess the effects of a single stressful event, they usually evaluated the changes a day or so after the stress occurred. What makes these new studies unique is that the researchers observed the results of a single stressful event at a delayed point in time after the threat ended.
In the first study, a team of researchers from India found that one stressful event did not immediately damage the amygdala of rats. However, fast forward 10 days and these animals suffered from increased anxiety and changes to the structure of their brains, especially the amygdala.
This is similar to what happens in humans who develop post-traumatic stress disorder (PTSD) days, weeks, or months after a traumatic event occurred.
Research has shown that stress causes new nerve connections called synapses to form in the amygdala. But until now the significance of these new nerve connections wasn’t known. However, in a follow-up study to the one investigating the delayed effects of stress on the amygdala, the researchers observed that these new nerve connections result in higher electrical activity in this region of the brain. This increased activity happened 10 days after a stressful event, and it occurs thanks to a molecule on nerve cells known as the N-Methyl-D-Aspartate Receptor (NMDA-R). When the researchers blocked NMDA-R during a stressful event, it prevented the formation of new synapses and the elevated electrical activity at these nerve connections.
The Indian researchers weren’t the first to investigate the long-term effects of short-lived stress on the brain. Another team of researchers from Italy published a study in the same month as the Indian investigation that showed one stressful event can cause long-term damage. In this rodent study, the scientists evaluated the effects of one episode of stress on the brain region known as the prefrontal cortex, which is involved in planning complex cognitive behavior, making decisions, and influencing the way we act in a social environment. The researchers had previously found that exposure to only 40 minutes of stress increased the release of glutamate, the primary excitatory neurotransmitter in the prefrontal cortex. Abnormally high glutamate levels can result in excessive excitation of the receiving nerve cell, resulting in anxious feelings and behavior.
In this current study, the scientists found that this increased release of glutamate doesn’t just stop when the stress is over. It continues for 24 hours after the stress stops.
During this time frame, dendrites in the prefrontal cortex that have receptors for glutamate experience atrophy.
Dendrites are extensions of nerve cells that act as the cells’ signal-transmitting telephone wires. This dendrite atrophy continued for two weeks after the stressful event, giving researchers a glimpse into how PTSD and other stress-related conditions may develop.
Stress Is Literally Killing Us
Whether you experience a life-shattering event or have to deal with the hassles of living in the modern world, there’s no doubt stress is a threat to your health. According to the latest Stress in America survey by the American Psychological Association, nearly one-third of adults report that stress has a very strong or strong impact on their physical and mental health.
Science paints a disturbing picture of the health consequences of stress. A recent study from Oregon State University found that older men who encounter a lot of stress—whether from everyday hassles such as battling traffic jams, job stress or arguments with family and friends or from significant life events such as the loss of a job or the death of a spouse—are more likely to die sooner compared with their less-stressed peers.
Stress is linked to most if not all the diseases of modern times, not the least of which is cardiovascular disease. In fact, the changes that occur in the amygdala after a stressful event may be to blame for the increased risk of cardiovascular disease in stressed people.
Researchers have found that activation of the amygdala increases immune system activity, which in turn increases the risk of having a heart attack or stroke.
They conducted two human studies. In the first, the team evaluated imaging and medical records from 293 people who had PET or CT scans to measure the activity of brain regions and also inflammation of the arteries. None of the participants had cancer or cardiovascular disease when the imaging was completed and medical record information on each subject was available from at least three additional visits to their doctors over the next two to five years.
Of the participants, 22 experienced a cardiovascular event such as a heart attack, stroke, or episode of angina during follow-up. The level of activity in the amygdala before the cardiovascular event was strongly linked to a greater risk of suffering a cardiovascular event. Even when the researchers controlled for traditional cardiovascular risk factors that association remained strong. And the association was even stronger between amygdala activity and major adverse cardiovascular events.
Amygdala activity also predicted the timing of cardiovascular events. People who had the most amygdala activity suffered from events sooner than those with the least amount of excess activity and this increase in events was related in part to increased arterial inflammation.
In the second study, 13 people with a history of PTSD received PET scans to determine amygdala activity and arterial inflammation. Their stress levels were strongly linked with both amygdala activity and arterial inflammation.
Stress Causes Depression
It is well known that stress causes depression. And new research has found that depression can actually cause the cerebral cortex of the brain to change shape, another indirect means by which stress can cause damage. Scientists have found that in humans, the cerebral cortex thickens in response to depression as a way to compensate for the depression, since the thicker the cortex, the fewer the symptoms. In depressed people treated with antidepressant drugs, the medications reduce the symptoms. This leads to the cortex becoming as thin as in non-depressed people.
Other Consequences of Stress
Stress impairs memory by blocking the formation of new nerve connections in the cerebral cortex. Stress also causes altered behaviors that further damage the brain. According to the Stress in America Survey, about two in five adults (39%) overate or ate unhealthy foods in the previous month because of stress. Researchers have observed that binge eating—a common habit in stressed people—changes the makeup of proteins in the brain and that this ability is driven by mutations in specific genes or a decrease in their activity.
Natural Solutions for Brain Recovery During Stress
Research indicates that a number of amino acids, vitamins and minerals can help restructure brain chemistry after experiencing chronic or short-term stress. These nutrients can support the function of the three brain areas affected by stress—the cerebral cortex, hippocampus and amygdala.
L-Theanine
L-theanine is the amino acid in green tea that gives the tea its flavor and is responsible for green tea’s beneficial effects. L-theanine promotes the generation of alpha brain waves, which are produced when the brain is in a relaxed state. Animal studies indicate L-theanine enhances levels of the neurotransmitters serotonin, dopamine and GABA, which are important for maintaining a good mood. In addition, it has been shown to block glutamate receptors, preventing excessive nerve cell excitation that would otherwise occur.
Its beneficial effects on neurotransmitters contribute to L-theanine’s ability to promote restful sleep. Moreover, L-theanine increased the effectiveness of the sedative midazolam in reducing anxiety when both were given together to mice. Additionally, following a stressful event, 200 mg of L-theanine increased relaxation.
L-theanine also influences gene expression in both the hippocampus and amygdala of rats, indicating it has beneficial effects on two areas of the brain affected by stress.
The genes affected by L-theanine are involved in reducing depression, stress enhanced fear learning, chronic mild stress, anxiety, mood disorders and psychoses.
In one randomized, double-blind, placebo-controlled study, 34 healthy adults consumed an L-theanine-based nutrient drink or a placebo and were exposed to a stressful situation that involved cognitive multitasking. L-theanine significantly reduced the participants’ stress response to this stressor one hour after administration of the L-theanine drink compared with the placebo. Levels of the stress hormone cortisol also were lower three hours after drinking the L-theanine.
L-theanine also blocks the NMDA receptor. Recall that the NMDA receptor is responsible for the increased activity that occurs in the amygdala after a stressful event. It is this activity that is linked to PTSD. In addition, activation of the NMDA receptor is associated with the buildup of amyloid-beta proteins, a hallmark of Alzheimer’s disease.
Tyrosine
Tyrosine is the precursor for the neurotransmitter norepinephrine, which is important for memory and is lowered during chronic stress. L-tyrosine stops the decline in norepinephrine that occurs during stressful situations. It also enhances mental and physical performance during stress. Supplementation with tyrosine boosts norepinephrine levels in the brains of mice, enhancing the animals’ learning and memory abilities.
Studies have found that supplementation with tyrosine improves the performance on tasks in people subjected to stress and fatigue.
It has improved performance in a variety of stressful situations including sleep deprivation, combat training, exposure to cold, and unpleasant background noise. For example, people exposed to long workdays exceeding 12 hours are often deprived of sleep and are fatigued. This results in increased stress and anxiety, a bad mood, and a decline in performance on tasks. Researchers investigated the effect of tyrosine on this scenario. The subjects were deprived of sleep from 7:30 p.m. to 8:20 a.m. the following day. They had been awake during the day prior to the study’s start. Six hours after their sleep deprivation started, the subjects were given tyrosine. Tyrosine was able to prevent declines in performance and vigilance and these benefits lasted about three hours.
Another group of researchers tested the effects of tyrosine on 21 cadets tackling a difficult military combat training course. Ten subjects were given five daily doses of a protein-rich drink containing two grams of tyrosine, and 11 subjects drank a carbohydrate rich drink with the same amount of calories. The tyrosine group performed better on tasks involving memory and tracking compared to subjects drinking the carbohydrate beverage. The tyrosine group also experienced a drop in systolic blood pressure.
L-Glutamine
The amino acid glutamine plays an important role as a precursor of neurotransmitters such as GABA. Levels of the amino acid glutamine drop sharply during illness and stress. Normal blood levels of glutamine are not enough to meet the body’s increased need for this nutrient when under stress and it is therefore critical that it be replenished.
Glutamine’s role in brain health is evident by the fact that glutamine supplementation to preterm infants results in increased white matter, hippocampus and brain stem volumes when the children reach school-age.
Additionally, glutamine levels are lower in brains of rodents with experimental Alzheimer’s disease. In cell culture studies, increasing glutamine levels in the medium protects neuronal cells against amyloid-beta. In mouse models of familial Alzheimer’s, 10 days of dietary glutamine supplementation blocked neuronal cell cycle activation and the damaging effects of tau, proteins that build up in the brain during Alzheimer’s disease.
5-HTP
The essential amino acid tryptophan is converted into 5-hydroxytryptophan (5-HTP), a precursor to the neurotransmitter serotonin. Supplementation with 5-HTP normalizes serotonin metabolism, enhances sleep and improves mood. In addition, 5-HTP counteracts the stimulant properties of amphetamine-type drugs, indicating it has a calming effect.
The dorsal raphe nucleus of the brain inhibits activity of neurons in the amygdala. As mentioned earlier, stress causes higher electrical activity in this region of the brain, which is in part responsible for the long-lasting effects of stress. When serotonin levels are low, however, this inhibitory effect of the dorsal raphe nucleus doesn’t occur. Researchers lowered the serotonin levels of animals then injected 5-HTP to boost levels of the neurotransmiter. Once this happened, the amygdala cells were again able to respond to stimulation by the dorsal raphe nucleus.
Through its ability to enhance serotonin levels, 5-HTP is also a cognitive enhancer. Increased brain serotonin activity is linked to improvements in cognitive function in animals and humans. On the other hand, decreasing brain serotonin levels through a tryptophan-deficient diet worsens cognitive function.
N-Acetyl L-Cysteine (NAC)
NAC is a precursor of the important antioxidant, glutathione. NAC resolves imbalances in the neurotransmitters glutamate and dopamine. It causes a reduction in glutamate release, which has been shown to stop drug-seeking behaviors in animals and in humans with substance use disorders. NAC improved social interaction and reduced anxiety-like behaviors in a rat model of autism by improving the function of the amygdala and the way that it uses glutamate. It had equally beneficial effects in a double-blind, randomized, placebo-controlled trial in children with autism. Compared with the placebo, NAC significantly reduced irritability in the children.
NAC is beneficial in conditions where stress leads to depression.
Rats exposed to 40 days of chronic mild stress were given either NAC or the combination of NAC and an iron-chelating drug. In the stressed rats treated with the saline control, sweet food intake decreased (a sign of stress in rats). NAC alone or in combination with the drug reversed this effect. NAC and the drug decreased levels of markers of free radical damage and increased activity of antioxidant enzymes in the prefrontal cortex, amygdala and hippocampus.
NAC’s ability to regulate glutamate levels makes it an agent of interest in studies investigating addictive disorders including gambling addictions. A review of the medical literature found evidence for the use of NAC in cocaine and marijuana addiction, bipolar disorder, depression, nail biting, skin picking, obsessive-compulsive disorder and schizophrenia. Moreover, NAC normalizes elevated glutamate levels in cocaine-dependent people. Higher glutamate levels are related to relapse.
Vitamins and Minerals
Combining the above nutrients with an array of vitamins, minerals, and antioxidants can further support the brain’s ability to recover and to improve mood after chronic and short-term stress. Vitamin B6 in the form of pyridoxal 5′ phosphate assists in the conversion of tryptophan to serotonin. Additionally, many people have a genetic defect that stops them from metabolizing folate effectively. This defect combined with low levels of vitamin B6 and B12 can lead to high levels of the amino acid homocysteine.
High levels of this toxic amino acid can reduce levels of the neurotransmitters dopamine, norepinephrine, epinephrine and serotonin, increasing the risk of developing depression.
Neurotransmitter generation in the cerebellum, hippocampus, and amygdala are all affected by folate deficiency.
Excess homocysteine also is converted into byproducts that activate the NMDA receptor. Remember that activation of the NMDA receptor is linked to the long-term effects of stress. A special type of folate known as L-methyl folate (L-MTHF) is more useable to the body than regular folate in people who have the genetic defect.
Vitamin C is another important anti-stress nutrient. A study in high school students found that this vitamin was effective in reducing stress. Another study of healthy young adults undergoing stress found that vitamin C reduced stressed feelings and lowered levels of blood pressure and the stress hormone cortisol.
Magnesium also possesses anti-stress properties. Stressful experiences cause more magnesium to be eliminated from the body. Deficiency of this mineral is related to a decreased ability to cope with and rebound from stress. Supplementation with magnesium, on the other hand, improves the nervous system’s ability to recover. This mineral blocks the NMDA receptor, indicating it may have beneficial effects on the amygdala during and after a stressful event.
Another mineral, chromium picolinate, may help conquer stress-related food cravings. Researchers have found that chromium can reduce the frequency of binge eating and symptoms of depression in people with binge eating disorder.
Conclusion
The stressful world we live in is taking its toll on our brains. Even just one stressful experience can cause long-term damage to important brain regions such as the amygdala. L-theanine, tyrosine, L-glutamine, 5-HTP, and NAC along with complementary vitamins, minerals and antioxidants may assist with brain recovery and reduce the worn-out feeling and bad mood that accompanies stress.
References:
- Yasmin F, Saxena K, McEwen BS, et al. “The delayed strengthening of synaptic connectivity in the amygdala depends on NMDA receptor activation during acute stress.” Physiol Rep 2016 Oct; 4(20): e13002.
- Musazzi L, Tornese P, Sala N, et al. “Acute stress is not acute: sustained enhancement of glutamate release after acute stress involves readily releasable pool size and synapsin I activation.” Molecular Psychiatry. Published online October 4 2016 doi:10.1038/mp.2016.175.
- American Psychological Association. http://www.apa.org/news/press/releases/stress/2015/highlights.aspx Accessed May 13, 2017.
- Aldwin CM, Jeong YJ, Igarashi H, et al. “Do hassles mediate between life events and mortality in older men?” Experimental Gerontology. 2014; DOI: 10.1016/j.exger.2014.06.019
- Tawakol A, Ishai A, Takx RA, et al. Relation between resting amygdalar activity and cardiovascular events: a longitudinal and cohort study. Lancet 2017;389(10071):834-45.
- Bansal R, Hellerstein DJ, Peterson BS. Evidence for neuroplastic compensation in the cerebral cortex of persons with depressive illness. Molecular Psychiatry. 7 March, 2017 doi:10.1038/mp.2017.34
- Saito S, Kimura S, Adachi N, et al. An in vitro reproduction of stress-induced memory defects: Effects of corticoids on dendritic spine dynamics. Sci Rep. 2016 Jan 14;6:19287.
- Kirkpatrick SL, Goldberg LR, Yazdani N, et al. Cytoplasmic FMR1-Interacting Protein 2 Is a Major Genetic Factor Underlying Binge Eating. Biological Psychiatry. 2017;81(9):757-69.
- Sampogna G, Del Vecchio V, Luciano M, et al [Should we continue to use benzodiazepines in clinical practice?]. [Article in Italian] Recenti Prog Med. 2015;106(6):243-5.
- Christiansen E, Agerbo E, Bilenberg N, et al. SSRIs and risk of suicide attempts in young people – A Danish observational register-based historical cohort study, using propensity score. Nord J Psychiatry. 2015 Aug 7:1-9. [Epub ahead of print.]
- Morrison J, Schwartz TL. Adolescent angst or true intent? Suicidal behavior, risk, and neurobiological mechanisms in depressed children and teenagers taking antidepressants. Int J Emerg Ment Health. 2014;16(1):247-50.
- Ferguson JM. SSRI Antidepressant Medications: Adverse Effects and Tolerability. Prim Care Companion J Clin Psychiatry. 2001;3(1):22-7.
- Alén F, Orio L, Gorriti MÁ. Increased alcohol consumption in rats after subchronic antidepressant treatment. Int J Neuropsychopharmacol. 2013;16(8):1809-18.
- Mawanda F, Wallace RB, McCody M, et al. PTSD, Psychotropic Medication Use, and the Risk of Dementia Among US Veterans: A Retrospective Cohort Study. Journal of the American Geriatrics Society. Feb 8, 2017. [Epub ahead of print.]
- Quaglio G, Lugoboni F, Fornasiero A, et al. Dependence on zolpidem: two case reports of detoxification with flumazenil infusion. Int Clin Psychopharmacol. 2005;20(5):285-7.
- Rappa LR, Larose-Pierre M, Payne DR, et al. Detoxification from high-dose zolpidem using diazepam. Ann Pharmacothe. 2004;38(4):590-4.
- Nathan PJ, Lu K, Gray M, et al. The neuropharmacology of L-theanine(N-ethyl-L-glutamine): a possible neuroprotective and cognitive enhancing agent. J Herb Pharmacother. 2006;6(2):21-30.
- Heese T, Jenkinson J, Love C, et al. Anxiolytic effects of L-theanine–a component of green tea–when combined with midazolam, in the male Sprague-Dawley rat. AANA J. 2009;77(6):445-9.
- Kimura R, Murata T. Effect of theanine on norepinephrine and serotonin levels in rat brain. Chem Pharm Bull (Tokyo). 1986;34(7):3053-7.
- Ceremuga TE, Martinson S, Washington J, et al. Effects of L-theanine on posttraumatic stress disorder induced changes in rat brain gene expression. ScientificWorldJournal. 2014;2014:419032. doi: 10.1155/2014/419032.
- White DJ, de Klerk S, Woods W, et al. Anti-Stress, Behavioural and Magnetoencephalography Effects of an L-Theanine-Based Nutrient Drink: A Randomised, Double-Blind, Placebo-Controlled, Crossover Trial. Nutrients. 2016;8(1). pii: E53. doi: 10.3390/nu8010053.
- Di X, Yan J, Zhao Y. L-theanine protects the APP (Swedish mutation) transgenic SH-SY5Y cell against glutamate-induced excitotoxicity via inhibition of the NMDA receptor pathway. Neuroscience. 2010;168(3):778-86.
- Wang Z, Li J, Wang Z, et al. L-tyrosine improves neuroendocrine function in a mouse model of chronic stress. Neural Regen Res. 2012;7(18):1413-9.
- Salter CA. Dietary tyrosine as an aid to stress resistance among troops. Mil Med. 1989;154(3):144-6.
- Neri DF, Wiegmann D, Stanny RR, et al. The effects of tyrosine on cognitive performance during extended wakefulness. Aviat Space Environ Med.1995;66:313-9.
- Deijen JB, Wientjes CJ, Vullinghs HF, et al. Tyrosine improves cognitive performance and reduces blood pressure in cadets after one week of a combat training course. Brain Res Bull.1999;48:203-9.
- Shurtleff D, Thomas JR, Schrot J, et al. Tyrosine reverses a cold-induced working memory deficit in humans. Pharmacol Biochem Behav.1994;47:935-41.
- Deijen JB, Orlebeke JF. Effect of tyrosine on cognitive function and blood pressure under stress. Brain Res Bull.1994;33:319-23.
- Meynial-Denis D. Glutamine metabolism in advanced age. Nutr Rev. 2016;74(4):225-36.
- de Kieviet JF, Vuijk PJ, van den Berg A, et al. Glutamine effects on brain growth in very preterm children in the first year of life. Clin Nutr. 2014;33(1):69-74.
- de Kieviet JF, Oosterlaan J, Vermeulen RJ, et al. Effects of glutamine on brain development in very preterm children at school age. Pediatrics. 2012;130(5):e1121-7.
- Chen J, Herrup K. Glutamine acts as a neuroprotectant against DNA damage, beta-amyloid and H2O2-induced stress. PLoS One. 2012;7(3):e33177.
- Leu-Semenescu S, Arnulf I, Decaix C, et al. Sleep and rhythm consequences of a genetically induced loss of serotonin. Sleep. 2010;33(3):307-14.
- Muszy?ska B, ?ojewski M, Rojowski J, et al. Natural products of relevance in the prevention and supportive treatment of depression. Psychiatr Pol. 2015; 49(3):435-53.
- Baumann MH, Williams Z, Zolkowska D, et al. Serotonin (5-HT) precursor loading with 5-hydroxy-l-tryptophan (5-HTP) reduces locomotor activation produced by (+)-amphetamine in the rat. Drug Alcohol Depend. 2011;114(2-3):147-52.
- Wang RY, Aghajanian GK. Inhibiton of neurons in the amygdala by dorsal raphe stimulation: mediation through a direct serotonergic pathway. Brain Res. 1977;120(1):85-102.
- Khaliq S, Haider S, Ahmed SP, et al. Relationship of brain tryptophan and serotonin in improving cognitive performance in rats. Pak J Pharm Sci. 2006;19(1):11-5.
- Olive MF, Cleva RM, Kalivas PW, et al. Glutamatergic medications for the treatment of drug and behavioral addictions. Pharmacol Biochem Behav. 2012; 100(4):801-10.
- Chen YW, Lin HC, Ng MC, et al. Activation of mGluR2/3 underlies the effects of N-acetylcystein on amygdala-associated autism-like phenotypes in a valproate-induced rat model of autism. Front Behav Neurosci. 2014;8:219.
- Hardan AY, Fung LK, Libove RA. A randomized controlled pilot trial of oral N-acetylcysteine in children with autism. Biol Psychiatry. 2012;71(11):956-61.
- Arent CO, Réus GZ, Abelaira HM, et al. Synergist effects of n-acetylcysteine and deferoxamine treatment on behavioral and oxidative parameters induced by chronic mild stress in rats. Neurochem Int. 2012;61(7):1072-80.
- Grant JE, Kim SW, Odlaug BL. N-acetyl cysteine, a glutamate-modulating agent, in the treatment of pathological gambling: a pilot study. Biol Psychiatry. 2007;62(6):652-7.
- Deepmala, Slattery J, Kumar N, et al. Clinical trials of N-acetylcysteine in psychiatry and neurology: A systematic review. Neurosci Biobehav Rev. 2015; 55:294-321.
- Schmaal L, Veltman DJ, Nederveen A, et al. N-acetylcysteine normalizes glutamate levels in cocaine-dependent patients: a randomized crossover magnetic resonance spectroscopy study. Neuropsychopharmacology. 2012;37(9):2143-52.
- Shabbir F, Patel A, Mattison C. Effect of diet on serotonergic neurotransmission in depression. Neurochem Int. 2013;62(3):324-9.
- Bhatia P, Singh N. Homocysteine excess: delineating the possible mechanism of neurotoxicity and depression. Fundam Clin Pharmacol. 2015;29(6):522-8.
- Jadavji NM, Wieske F, Dirnagl U, et al. Methylenetetrahydrofolate reductase deficiency alters levels of glutamate and γ-aminobutyric acid in brain tissue. Mol Genet Metab Rep. 2015;3:1-4.
- de Oliveira IJ, de Souza VV, Motta V, et al. Effects of Oral Vitamin C Supplementation on Anxiety in Students: A Double-Blind, Randomized, Placebo-Controlled Trial. Pak J Biol Sci. 2015;18(1):11-8.
- Brody S, Preut R, Schommer K, et al. A randomized controlled trial of high dose ascorbic acid for reduction of blood pressure, cortisol, and subjective responses to psychological stress. Psychopharmacology (Berl). 2002;159(3):319-24.
- Tarasov EA, Blinov DV, Zimovina UV, et al. [Magnesium deficiency and stress: Issues of their relationship, diagnostic tests, and approaches to therapy].[Article in Russian.] Ter Arkh. 2015;87(9):114-22.
- Wilder-Smith CH, Knöpfli R, Wilder-Smith OH. Perioperative magnesium infusion and postoperative pain. Acta Anaesthesiol Scand. 1997;41(8):1023-7.
- Brownley KA, Von Holle A, Hamer RM, et al. A double-blind, randomized pilot trial of chromium picolinate for binge eating disorder: results of the Binge Eating and Chromium (BEACh) study. J Psychosom Res. 2013;75(1):36-42.












[…] fonte: https://nutritionreview.org/2017/11/protecting-the-brain-from-the-damaging-effects-of-modern-stress-… […]
[…] fonte: https://nutritionreview.org/2017/11/protecting-the-brain-from-the-damaging-effects-of-modern-stress-… […]